"*" indicates required fields
This online course consists of four modules as described and illustrated below.
500: Electronic data integrity in the Life Sciences industry (1 hour)
Module 500
This module aims to:
510: Computer system validation (1 hour)
Module: 510
This module aims to:
The learning objectives of this module are that you will be able to:
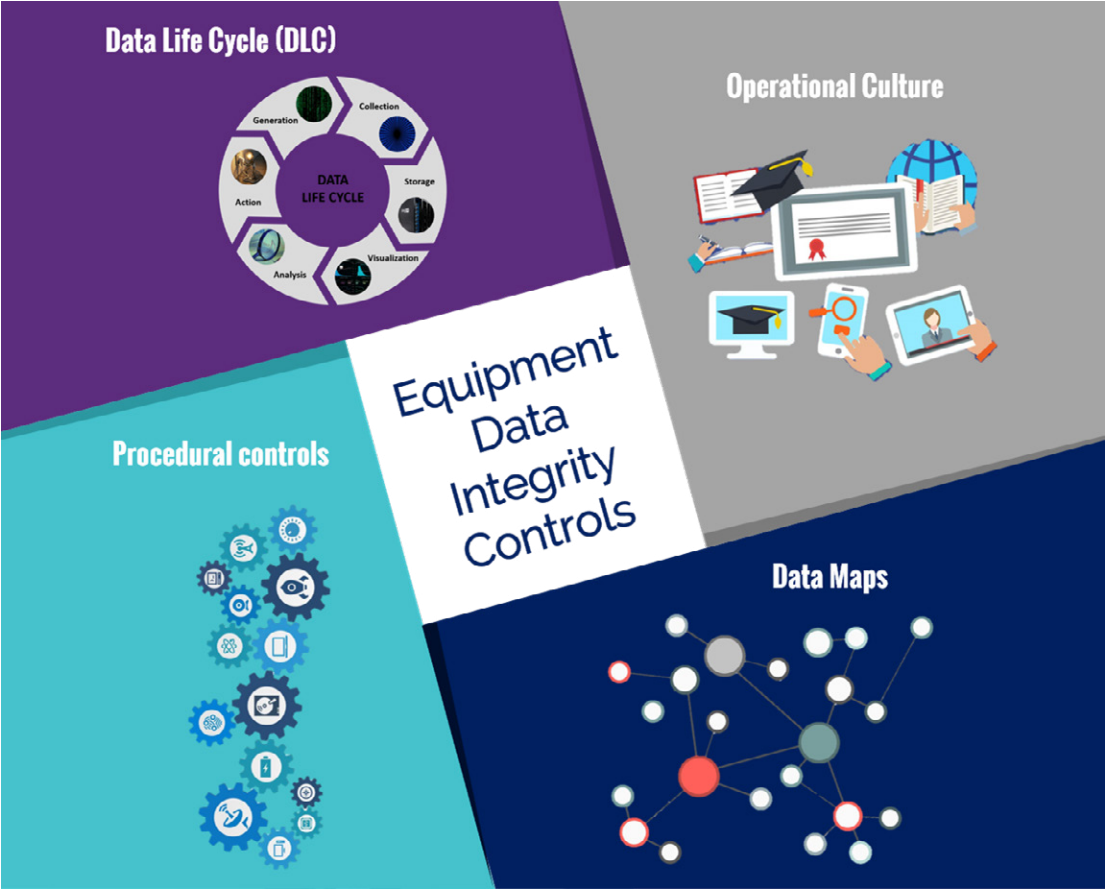
520: Equipment considerations for data integrity (1 hour)
Module: 520
This module aims to:
The learning objectives for this module will allow you to:
Module 530: Application Types for Data Integrity (1 hour)
Module: 530
This module aims to:
The learning objectives of this module are that you will be able to:
“The wide breadth of content offered by the IAAE® yields an opportunity to increase capability across a commensurately diverse set of functional groups and raise the tide for all boats.”
- Dan Kim, Director, Automation & Digital Operations, PSC Biopharmaceuticals with GSK